FDA’s Detection of Banned Antibiotics in Indonesian Shrimp Continues in 2026
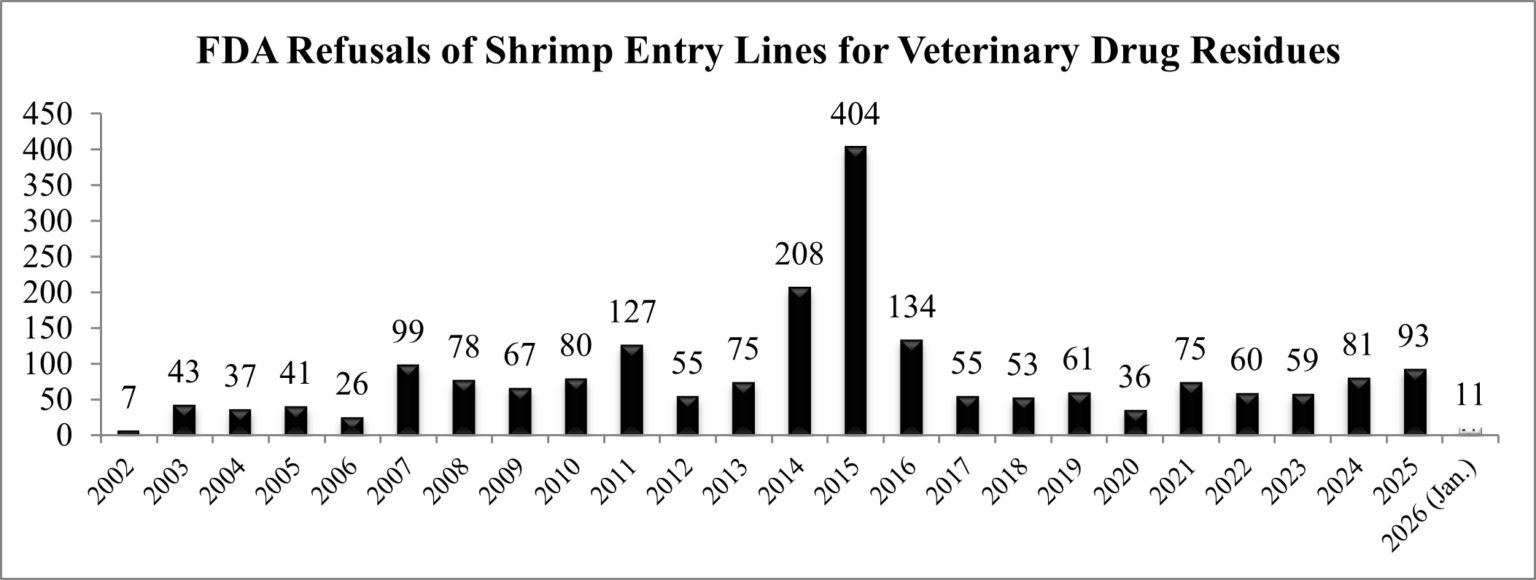
As shown in the chart below, the FDA’s reporting of shrimp entry line refusals for reasons related to banned antibiotics continues the trend of increasing rejections over the last three years.
As explained in more detail below, seven of the entry line refusals in January were of shrimp from Indonesia, continuing the FDA’s response to the Indonesian shrimp aquaculture’s industry adoption of banned antibiotics. To put the seven entry line refusals of Indonesian shrimp in January 2026 in context, over the nine-year period spanning from 2016 through 2024, the FDA refused a grand total of eight Indonesian shrimp entry lines for reasons related to veterinary drug residues.
The eleven (11) reported entry lines of shrimp refused for veterinary drug residues in January were attributed to shipments from four different exporters in Hong Kong, Indonesia, and Vietnam. As noted below, three of these four exporters are Best Aquaculture Practices (BAP)-certified shrimp processors:
PT. Tamron Akuatik Produk Industri (Indonesia), a company that currently operates under a four-star BAP certification for its processing plant (P10704) and that was added to Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) on August 28, 2025 for the presence of nitrofurans in its shrimp, had one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Southeast Imports on January 7, 2026; one entry line refused for breaded shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Southwest Imports on January 9, 2026; one entry line refused for breaded shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Southeast Imports on January 15, 2026; one entry line refused for breaded shrimp contaminated with nitrofurans by the Division of Northeast Imports on January 20, 2026; one entry line refused for breaded shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Northern Border Imports on January 23, 2026; one entry line refused for breaded shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Northeast Imports on January 27, 2026; and one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Southeast Imports on January 28, 2026;
Gallant Dachan Seafood Co., Ltd. (Vietnam), a company that currently operates under a three-star processor BAP certification (P10436) and is not currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”), Import Alert 16-127 (“Detention Without Physical Examination of All Seafood Due to Chloramphenicol”), or Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”), had two entry lines refused for breaded shrimp contaminated with nitrofurans by the Division of Southeast Imports on January 2, 2026;
Seavina Joint Stock Company (Vietnam), a company that currently operates under a four-star processor BAP certification (P10387) and is not currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”), Import Alert 16-127 (“Detention Without Physical Examination of All Seafood Due to Chloramphenicol”), or Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”), had one entry line refused for breaded shrimp contaminated with nitrofurans by the Division of Southeast Imports on January 21, 2026; and
Hoptai Marine Products Company Limited (China), a company that is not currently included on the “green” list for Import Alert 16-131 (“Detention Without Physical Examination of Aquacultured Shrimp, Dace, and Eel from China and Hong Kong SAR – Presence of New Animal Drugs and/or Unsafe Food Additives”), had one entry line refused for shrimp contaminated with veterinary drug residues by the Division of Northeast Imports on January 30, 2026.
In addition to these rejects, the FDA’s Division of West Coast Imports reported refusing an entry line of Vietnamese shrimp manufactured by Fimex VN for “ADDED BULK” on January 6th after sampling the shipment.
Soure: shrimpalliance
AQUA MINA CO., LTD
– Address: 685 Le Duc Anh Street, Quarter 39, Binh Hung Hoa Ward, Ho Chi Minh City
– Phone: 1800 6071 (Toll-free hotline)
– Email: sales@aquamina.com.vn or oversea@aquamina.com.vn
– Aqua Mina’s Official Distributor in Japan: REX INDUSTRIES CO., LTD
– Address: 1-9-3 Hishiya-Higashi, Higashi-Osaka 578-0948, JAPAN
– Email: kimakubo@rexind.co.jp
– Phone: +81-(0)72-961-9893
– Website: www.rexind.co.jp/e/

WE WORK FOR THE SUCCESS
Ngày đăng : 22/02/2026
1154 View
Other Articles
AQUA MINA | CELEBRATING THE 67TH ANNIVERSARY OF VIETNAM’S TRADITIONAL FISHERIES DAY
Restructuring seed production to enhance the competitiveness of the aquaculture industry
AQUA MINA COMMITS TO NO PRICE INCREASE – 15% OFF PVC Knife Gate Valves, Double Flanges, and Elbow Flanges TO SUPPORT SHRIMP AND FISH FARMERS
Global shrimp prices – Week 12: Ecuador shrimp prices recover, while India and Indonesia remain stable
The global surimi market is projected to grow strongly.
Cà Mau: Expanding the RAS-IMTA model toward sustainable, zero-discharge shrimp farming.
From seed stock to farming equipment, high-tech aquaculture attracts strong interest in Ho Chi Minh City
Shrimp industry enterprises proactively adapt to global fluctuations
DAY 3 – AQUA MINA | STRONG CONNECTIONS – A MEMORABLE CLOSING AT VIETSHRIMP AISA 2026
DAY 2 – AQUA MINA | DEEP INSIGHTS & SMART SOLUTIONS FOR MODERN AQUACULTURE
DAY 1 – AQUA MINA | A POWERFUL OPENING AT VIETSHRIMP ASIA 2026
OFFICIAL OPENING of the International-Scale Aquaculture Exhibition Series | See You Again with AQUA MINA at VietShrimp Asia 2026



















.jpg)